Vyndamax (tafamidis) for ATTR-CM
What is Vyndamax for ATTR-CM?
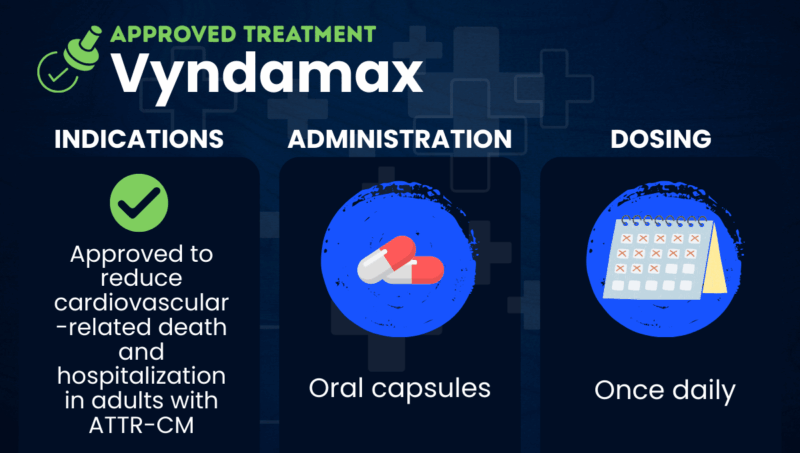
Vyndamax (tafamidis) is an oral transthyretin stabilizer used to reduce hospitalization and death related to cardiovascular disease in adults with wild-type or hereditary transthyretin amyloid cardiomyopathy (ATTR-CM).
ATTR-CM occurs when the protein transthyretin becomes unstable, causing its typical four-part structure to break down into individual components that are prone to misfolding and clumping together. These clumps, known as amyloid deposits, accumulate in the heart and cause damage.
Vyndamax works to stabilize the four-part transthyretin structure so it won’t break down. This prevents the formation of new amyloid deposits in the heart, thereby helping to stop or slow the progression of ATTR-CM.
The medication is marketed by Pfizer and taken as a once-daily oral capsule. It is sold in the European Union under the brand name Vyndaqel.
Therapy snapshot
| Brand name | Vyndamax |
| Chemical name | Tafamidis |
| Usage | Used to reduce cardiovascular hospitalization and mortality in adults with ATTR-CM |
| Administration | Oral capsules |
Who can take Vyndamax?
Vyndamax is approved in the U.S. for the treatment of adults with wild-type or hereditary ATTR-CM. It is designed to reduce cardiovascular-related hospitalization and death.
There are no known contraindications for its use.
The medication is approved for treating ATTR-CM in other regions, including the EU. There, a different formulation of tafamidis is also approved for a related condition called transthyretin amyloidosis with polyneuropathy.
How is Vyndamax administered?
Vyndamax is available in capsule form, taken orally once daily. The recommended daily dose is a single, 61 mg capsule.
The capsules should be swallowed whole and not crushed or cut.
Vyndamax in clinical trials
Another formulation of Vyndamax’s active ingredient, tafamidis, was initially approved in the U.S. under the brand name Vyndaqel, which has since been discontinued. Phase 1 data in healthy volunteers showed that Vyndaquel (80 mg) was bioequivalent to Vyndamax (61 mg), indicating that it resulted in the same amount of tafamidis in the body and, therefore, would be expected to have similar clinical effects.
As such, Vyndamax’s regulatory approval was largely supported by a Phase 3 study of Vyndaqel called ATTR-ACT (NCT01994889). The study enrolled 441 adults with ATTR-CM who were randomly assigned to receive Vyndaqel (20 or 80 mg) or a placebo daily for 30 months, or 2.5 years, in addition to standard care. The results showed that, compared with the placebo, Vyndaqel:
- resulted in significantly lower rates of death from any cause and hospitalization due to cardiovascular problems
- slowed decline in exercise capacity, measured as the distance walked in six minutes
- slowed declines in life quality, with more people on the medication reporting stabilizations or improvements in health-related life quality, heart failure symptoms, and general health
Trial participants could then enter into a long-term extension study (NCT02791230), in which all would receive active treatment for nearly five years. All participants started on Vyndaqel but eventually switched to Vyndamax.
The results showed that individuals who had started on Vyndaqel in the main trial — meaning they had begun active treatment sooner and been on it for longer — had higher long-term survival rates than those who had switched from the placebo.
Vyndamax side effects
The U.S. prescribing label does not list any common side effects of Vyndamax. In clinical trials, most side effects occurred at similar rates in the Vyndamax and placebo groups, so it could not be established that they were related to the treatment.
Vyndamax could cause harm to a developing fetus, and patients are advised to tell their healthcare provider right away if they are pregnant or think they may be pregnant. Patients are also advised not to breastfeed during treatment with Vyndamax.
Amyloidosis News Today is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by