Nexiguran ziclumeran for hATTR-PN
What is nexiguran ziclumeran for hATTR-PN?
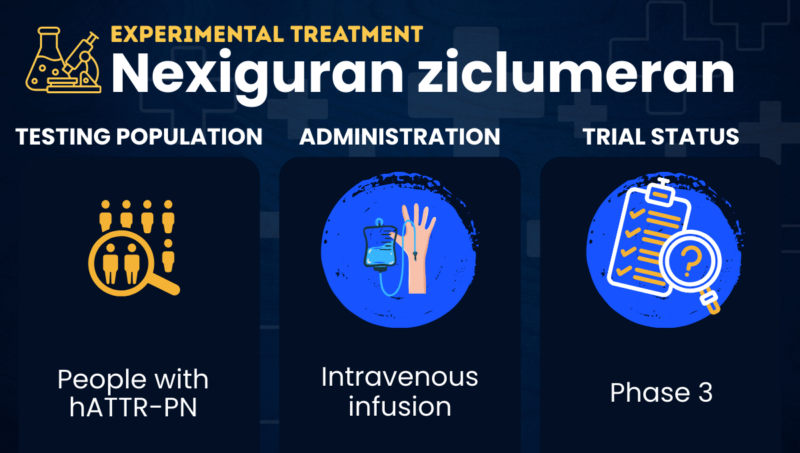
Nexiguran ziclumeran or nex-z for short (also known as NTLA-2001) is a gene-editing therapy being tested as a possible treatment for hereditary transthyretin amyloidosis with polyneuropathy (hATTR-PN).
In hATTR-PN, mutations in the TTR gene result in the production of an abnormal version of the transthyretin protein. This protein then harmfully accumulates in the body, particularly in the peripheral nerves, which are the nerve cells outside the brain and spinal cord.
Nex-z uses CRISPR/Cas9 gene-editing technology to inactivate the TTR gene in the liver, where most transthyretin is made. By lowering transthyretin production, nex-z is expected to reduce levels of the toxic protein clumps that cause nerve damage in hATTR-PN. It is designed to be given as a one-time intravenous, or into-the-vein, infusion.
The therapy, now in Phase 3 testing for hATTR-PN, is being developed by Intellia Therapeutics in collaboration with Regeneron Pharmaceuticals. It’s also in clinical trials as a possible treatment for transthyretin amyloid cardiomyopathy (ATTR-CM), a related disease in which transthyretin mainly accumulates in the heart.
Nex-z has been granted orphan drug status in the U.S. and the European Union, as well as regenerative medicine advanced therapy status in the U.S. Both are designations intended to facilitate a therapy’s clinical development.
Therapy snapshot
| Treatment name | Nexiguran ziclumeran |
| Administration | One-time intravenous infusion |
| Clinical testing | In Phase 3 testing for hATTR-PN |
How will nexiguran ziclumeran be given in hATTR-PN?
In a Phase 3 trial, people with hATTR-PN received a single intravenous infusion of nex-z at a dose of 55 mg.
Nexiguran ziclumeran in hATTR-PN clinical trials
A Phase 3 trial called MAGNITUDE-2 (NCT06672237) is evaluating the safety and efficacy of nex-z versus a placebo in about 60 adults with hATTR-PN. The main goals are to evaluate changes in blood levels of transthyretin and symptoms of nerve damage (neuropathy) after about 1.5 years.
MAGNITUDE-2 was paused in October 2025 when a person with ATTR-CM who received nex-z in a separate clinical trial developed life-threatening liver damage and eventually died. The study resumed in January 2026 with new safety measures in place.
A previous Phase 1 clinical trial (NCT04601051) assessed the safety and effectiveness of a single nex-z infusion in adults with hATTR-PN or ATTR-CM. Various doses were evaluated. Results from the participants with hATTR-PN showed that nex-z led to:
- sustained reductions in TTR levels, by about 90%, that lasted for up to three years
- clinically meaningful reductions in neuropathy symptoms for 72% of participants with available data after two years
- stabilizations or improvements in neuropathy-related disability for most participants
- reductions in biomarkers of nerve damage and improvements in quality of life
Participants from that trial are now being monitored for as long as 15 years in a follow-up study (NCT05697861). That long-term study mainly aims to track safety.
Nexiguran ziclumeran side effects
In the Phase 1 trial, the most common side effects among hATTR-PN patients that were considered possibly related to nex-z included:
- infusion-related reactions
- headache
- decreased blood levels of thyroxine, a thyroid hormone
Amyloidosis News Today is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by