Attruby (acoramidis) for ATTR-CM
What is Attruby for ATTR-CM?
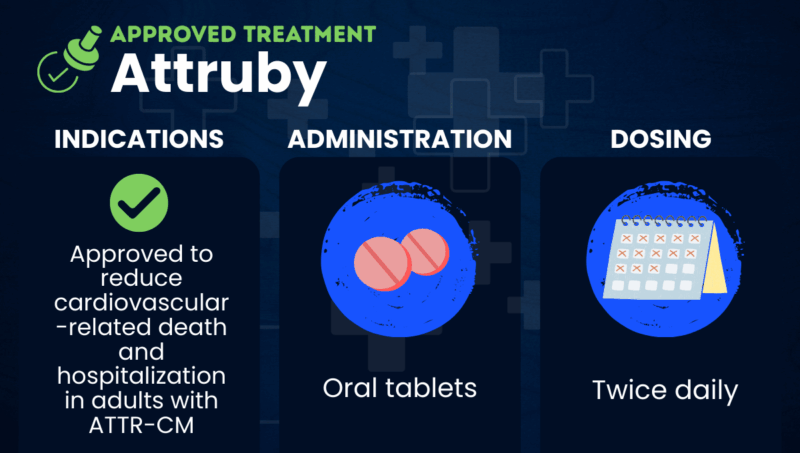
Attruby (acoramidis) is an oral transthyretin stabilizer approved to reduce cardiovascular-related hospitalization or death in adults with hereditary or wild-type transthyretin amyloid cardiomyopathy (ATTR-CM).
The protein transthyretin normally binds together in groups of four to perform its usual functions. But in ATTR-CM, it becomes unstable, and these functional units break down into individual parts. These then tend to misfold and accumulate in the heart, damaging it.
The disease can be caused by mutations in the TTR gene, which contains instructions for cells to produce transthyretin; it’s then known as hereditary ATTR-CM. It can also be caused by age-related cellular changes, in which case it’s known as wild-type ATTR-CM.
Like other TTR stabilizers, Attruby aims to support the four-part structure of transthyretin, preventing its breakdown and harmful accumulation. It is designed for enhanced potency by mimicking the effects of a naturally occurring TTR mutation that does not cause ATTR-CM but instead has protective, stabilizing effects on the TTR protein.
Bridgebio developed Attruby and is responsible for its U.S. commercialization. In the European Union, the medication is sold by Bayer under the brand name Beyonttra.
Therapy snapshot
| Brand name | Attruby |
| Chemical name | Acoramidis |
| Usage | Used to reduce cardiovascular-related mortality and hospitalization in adults with ATTR-CM |
| Administration | Oral tablets |
Who can take Attruby?
Attruby is approved in the U.S. to reduce cardiovascular death and cardiovascular-related hospitalization in adults with hereditary or wild-type ATTR-CM. There are no known contraindications for its use.
The medication is also indicated for adults with ATTR-CM in the EU.
How is Attruby administered?
Attruby is administered in the form of oral tablets. The recommended dosage is 712 mg (two tablets), taken twice daily, with or without food.
The tablets should be swallowed whole and should not be cut, crushed, or chewed.
Attruby in clinical trials
The primary clinical trial supporting the approval of Attruby in the U.S. was a Phase 3 study called ATTRibute-CM (NCT03860935). In the trial, more than 600 adults with wild-type or hereditary ATTR-CM were randomly assigned to receive a placebo or Attruby twice daily for 30 months, or 2.5 years.
The results showed benefits with treatment compared with the placebo:
- Treatment with Attruby led to significantly better clinical outcomes, evaluated using a metric that combined death from any cause, hospitalization related to cardiovascular problems, a molecular marker of heart damage, and changes in exercise capacity.
- The medication’s use led to higher blood transthyretin levels, suggesting an increase in protein stability, with larger early increases associated with higher survival rates.
- Attruby treatment tended to improve measures of heart structure and function.
After the study, ATTRibute-CM participants could enter an ongoing open-label extension study (NCT04988386) in which all are given Attruby.
Interim results after one year in the extension study showed that participants who had been on Attruby continuously since the start of ATTRibute-CM had significantly better long-term clinical outcomes than those who had switched from the placebo and thus started active treatment later. Attruby was also found to be similarly effective for hereditary and wild-type ATTR-CM.
A Phase 3 trial now underway, called ACT-EARLY (NCT06563895), is evaluating whether Attruby can prevent or delay the development of hereditary ATTR-CM or a related condition affecting the nerves, known as hereditary transthyretin amyloidosis with polyneuropathy. It enrolled individuals who have predisposing TTR gene mutations but don’t yet have any symptoms.
Attruby side effects
Side effects of Attruby may include gastrointestinal problems, such as diarrhea or abdominal pain. In clinical trials, these were typically mild and temporary.
Starting Attruby may cause reversible changes in blood tests related to kidney function.
Attruby may interact with certain medications, requiring more careful monitoring. Patients should tell their healthcare provider about all medications they are using.
Amyloidosis News Today is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by