Amvuttra (vutrisiran) for ATTR-CM
What is Amvuttra for ATTR-CM?
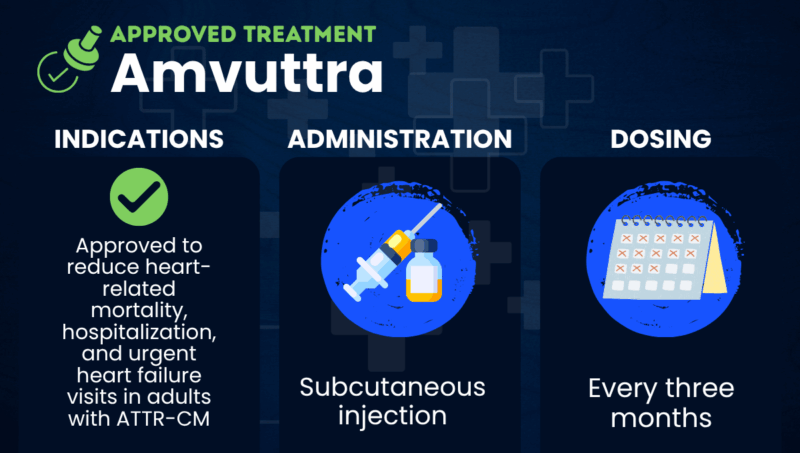
Amvuttra (vutrisiran) is an injectable therapy approved to reduce heart-related mortality, hospitalizations, and urgent heart failure visits in adults with transthyretin amyloid cardiomyopathy (ATTR-CM).
In ATTR-CM, the transthyretin protein forms abnormal clumps called amyloid deposits that accumulate in the body’s cells — mainly in heart tissue — leading to heart damage and related symptoms. This can be caused by mutations in the TTR gene, which encodes transthyretin, resulting in the production of an abnormal version of the protein (hereditary), or by age-related processes that cause normal transthyretin to become abnormal over time (wild-type).
A second-generation gene-silencing therapy, Amvuttra targets the TTR gene to reduce the amount of transthyretin that is produced. It contains a genetic fragment called a small interfering RNA (siRNA) that binds to and promotes the destruction of an intermediate template molecule needed for cells to produce transthyretin from the TTR gene. In doing so, Amvuttra helps prevent the accumulation of amyloid deposits in both hereditary and wild-type ATTR-CM, potentially slowing or stopping disease progression.
The therapy is given via under-the-skin (subcutaneous) injections and is marketed by Alnylam Pharmaceuticals. It is also approved for treating hereditary transthyretin amyloidosis with polyneuropathy (hATTR-PN), a related condition where amyloid deposits accumulate primarily in certain nerve cells.
Therapy snapshot
| Brand name | Amvuttra |
| Chemical name | Vutrisiran |
| Usage | Used to reduce heart-related hospitalizations, mortality, and urgent heart failure visits in adults with ATTR-CM |
| Administration | Subcutaneous injection |
Who with ATTR-CM can take Amvuttra?
Amvuttra is approved in the U.S. for treating adults with hereditary or wild-type ATTR-CM, to reduce cardiovascular mortality, cardiovascular hospitalizations, and urgent heart failure visits. The therapy is similarly approved in the European Union.
There are no known contraindications for its use.
How is Amvuttra administered in ATTR-CM?
Amvuttra is administered by a healthcare professional as a subcutaneous injection into the abdomen, thighs, or upper arms. The recommended dose is 25 mg once every three months, or four doses per year. Each injection can be completed in a few minutes.
Amvuttra in ATTR-CM clinical trials
Amvuttra’s approvals for ATTR-CM were supported by the Phase 3 HELIOS-B trial (NCT04153149), which involved 654 adults with hereditary or wild-type ATTR-CM who had a clinical history of heart failure. Participants received subcutaneous injections of either Amvuttra or a placebo every three months for 36 months (about three years). Data showed that:
- Amvuttra reduced the risk of heart-related hospitalizations, urgent heart failure visits, or death from any cause by 28% compared with the placebo, with similar benefits observed in the subset of patients who were not simultaneously using Vyndamax (tafamidis).
- Metrics of quality of life and exercise capacity improved more with Amvuttra than with the placebo.
- The treatment had favorable effects on cardiac structure and function relative to the placebo, with those given Amvuttra also tending to see stabilization or improvement in biomarkers of heart damage.
- Amvuttra was effective regardless of age or initial heart disease severity, although the greatest benefit was seen when treatment was initiated in earlier disease stages.
After the main part of the study, all the participants could receive Amvuttra for two more years in an open-label extension (OLE) period. Data showed that the benefits of Amvuttra on survival and cardiovascular events were further improved with up to six months of treatment in the OLE.
After completing two years in the OLE, participants can opt to enter another ongoing open-label extension study (NCT06679946), where all are receiving Amvuttra for up to three more years.
A separate Phase 3 trial called HELIOS-A (NCT03759379) tested Amvuttra in people with hATTR-PN. While the study mainly focused on nerve-related symptoms, the data also showed that Amvuttra improved heart-related measures in these patients.
Amvuttra side effects
The most common side effects of Amvuttra are:
- pain in the extremities (arms, legs, hands, feet)
- joint pain
- shortness of breath
- reduction in vitamin A levels
A doctor may recommend vitamin A supplements during Amvuttra treatment. Vitamin A deficiency can lead to a variety of symptoms, including night blindness and other vision problems. If such symptoms occur, patients may be referred to an eye specialist.
Amyloidosis News Today is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by